Cynapsus Therapeutics Announces Positive Phase 3 Dose Titration Phase Results with APL-130277 for the Treatment of OFF Episodes in Patients with Parkinson's Disease
-- Webcast with accompanying slides to discuss these findings tomorrow at 7:30 a.m. Eastern time
Top-line CTH-300 results expected in late Q3 or early Q4 --
TORONTO, July 18, 2022 (GLOBE NEWSWIRE) -- Cynapsus Therapeutics, Inc. (NASDAQ:CYNA) (TSX:CTH), a specialty central nervous system (CNS) pharmaceutical company developing and preparing to commercialize a fast-acting, easy-to-use, sublingual thin film for the on-demand management of debilitating OFF episodes associated with Parkinson’s disease (PD), today announced positive open-label dose titration phase (DTP) results from the ongoing CTH-300 pivotal Phase 3 efficacy and safety trial (CTH-300).
CTH-300 enrollment was completed on June 30, 2022 with 102 PD patients entering open-label DTP. The DTP part of CTH-300 is now complete, with data available from the first 92 patients. Remaining patient data will be entered upon completion of their first post-randomization visit.
Preliminary CTH-300 open-label DTP highlights include:
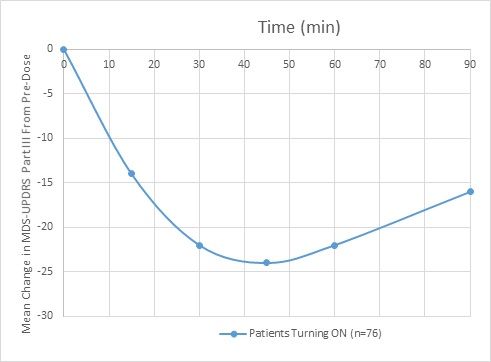
- The mean change in Movement Disorder Society’s Unified Parkinson’s Disease Rating Scale Part III (MDS-UPDRS Part III) from baseline to 30 minutes was 22 points
- In 60% of patients, improvement in motor function was reported between 5 and 12 minutes after dosing.
- Improvement in motor function was maintained beyond 90 minutes with a mean improvement of the MDS UPDRS Part III of 16 points at that time point
- 83% of patients entering the open-label dose titration phase turned from OFF to fully ON
- 78% of patients entering the open-label dose titration phase turned fully ON within 30 minutes; approximately 38% were fully ON at 15 minutes
- The median dose of APL-130277 turning patients to fully ON was 20mg
- APL-130277 was well tolerated. During the dose titration period: 16% of patients reported nausea that was mostly mild in intensity; 8% of patients reported dizziness; 4% of patients reported somnolence; 2% of patients reported vomiting; and 1% of patients reported symptomatic hypotension
- There were no reports of local irritation during the open-label DTP of the CTH-300 study
- A total of 16 patients were dosed and did not complete the DTP of the study; five (5) of such patients discontinued due to an adverse event, nine (9) of such patients discontinued because the highest dose in this study was not sufficient to turn them from OFF to ON within 45 minutes and two (2) of such patients discontinued for administrative reasons.
“Top-line results from the DTP of CTH-300 corroborate our Phase 2 findings supporting the potential benefit of APL-130277 as an on-demand therapy for the management of OFF episodes in patients with Parkinson’s disease who suffer motor fluctuations,” said Dr. Albert Agro, Chief Medical Officer of Cynapsus. “The robustness of this preliminary clinical data, particularly the 22-point improvement in MDS-UPDRS Part III, gives us confidence that our sample size of 80 patients randomized, is more than sufficient to show statistical and clinical meaningful changes at 90% power in our primary and key secondary clinical endpoints.”
Figure 1 (below) demonstrates the change from baseline of the MDS-UPDRS Part III score at 15, 30, 45, 60 and 90 minutes post-dosing^
|
|||
^Data captured is based on patients existing in the database as of July 15, 2022
Table 1 Illustrates the most common dopaminergic Adverse Events (AE) seen in CTH-300 Dose Titration Phase (n=92)*
| N=92 | Number of Patients with an Adverse Event | |||
| Any AE | Mild AE | Moderate AE | Severe AE | |
| N (%) | N (%) | N (%) | N (%) | |
| Nausea | 15 (16) | 12 (13) | 2 (2) | 1 (1) |
| Dizziness | 7 (8) | 4 (4) | 3 (3) | 0 (0) |
| Somnolence | 4 (4) | 3 (3) | 1 (1) | 0 (0) |
| Yawning | 3 (3) | 3 (3) | 0 (0) | 0 (0) |
| Vomiting | 2 (2) | 1 (1) | 1 (1) | 0 (0) |
| Symptomatic Hypotension | 1 (1) | 1 (1) | 0 (0) | 0 (0) |
| Stomatitis/ Irritation | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
* Data not final and subject to change
“The results seen in the open label dose titration period of CTH-300 are an important step in confirming earlier data that demonstrated that APL-130277 can provide PD patients with a safe, tolerable and rapid improvement in motor function during OFF episodes,” said Anthony Giovinazzo, President and CEO of Cynapsus. “We look forward to the completion of CTH-300 and preparing the overall study results for our NDA submission. APL-130277 is being developed to address a significant unmet need facing people with Parkinson’s disease. The preliminary results from CTH-300 DTP lead us to believe that APL-130277 may be able to fulfill the unmet needs of Parkinson’s patients seeking to better manage their motor fluctuations.”
These Phase 3 dose titration results are only a snap shot and the final data and results from the Phase 3 study, when released, could differ from such results.
Phase 3 Program Study Design
The CTH-300 study is a double-blind, placebo-controlled, parallel-design, two-part study. The study enrolled eligible PD patients and 102 patients entered the Open Label DTP. In the DTP, patients arrived to the clinic in the practically defined OFF state (withholding their morning dose of PD medications) and received the lowest dose of APL-130277 (10mg). Patients were assessed to determine if successful conversion from OFF to full ON occurred. If a full ON was not achieved, patients returned to the clinic on subsequent visits and received a higher dose of APL-130277 (at 5mg increments, up to 35mg) until the effective conversion dose was identified. Those patients that turned fully ON with a dose of APL-130277 during the DTP proceeded to the dose randomization part of the study where they were randomized to receive either the effective dose or placebo. The objective of the second part of CTH-300 is to evaluate the efficacy, safety and tolerability of APL-130277 versus placebo in patients with PD over a 12-week period. CTH-300 is well powered to demonstrate statistical significant reduction in MDS-UPDRS Part III with 80 patients randomized. A sample size of 80 randomized patients allows for greater than 90% power to achieve statistical significance at the primary and key secondary endpoint. The primary endpoint is mean change in the MDS-UPDRS Part III score at 30 minutes after 12 weeks of at-home dosing.
The CTH-301 Phase 3 Safety Study (CTH-301) is a long-term open-label, single arm safety study in PD patients who have at least one OFF episode per day, with total OFF time of at least two hours per day. The objective is to evaluate the safety and tolerability of APL-130277 in up to 200 patients with PD over a six-month period. Patients completing CTH-300 are eligible to enter CTH-301. In addition, de novo patients from approximately 65 sites in North America and Europe will be enrolled in CTH-301.
Data from CTH-300 and CTH-301 are expected to form the basis for the registration package necessary for a 505(b)(2) New Drug Application (NDA) with the U.S. Food and Drug Administration expected to be submitted in the first half of 2017.
About Parkinson’s Disease and OFF Episodes
More than 1 million people in the U.S. and an estimated 4 to 6 million people worldwide suffer from PD. It is a chronic, progressive neurodegenerative disease characterized by motor symptoms, including tremor at rest, rigidity and impaired movement, as well as significant non-motor symptoms, including cognitive impairment and mood disorders. PD’s prevalence is increasing with the aging of the population. OFF episodes are a complication of the disease. An estimated one quarter to one half of all people with PD whose symptoms are otherwise managed with ongoing drug therapy experience OFF episodes at least once daily and up to six times daily, with each episode lasting between 30 and 120 minutes.
About Apomorphine
Apomorphine is the only molecule approved for acute, intermittent treatment of OFF episodes for advanced PD patients, but is currently only approved as a subcutaneous injection in the United States, which can be painful and difficult to administer, particularly while suffering an OFF episode.
Cynapsus’ APL-130277 product candidate is a sublingual formulation of apomorphine for the treatment of OFF episodes. If approved, APL‐130277 will provide patients with a convenient and easy alternative to multiple daily injections.
Conference Call and Webcast
Cynapsus management will host an analyst meeting and live webcast with accompanying slides to discuss these findings tomorrow at 7:30 a.m. Eastern time. To view the live webcast, visit the Investor Relations section of the Company’s website at www.cynapsus.ca.
A replay of the webcast will be available 2 hours after completion of the meeting, and archived for 90 days on the Company's website at www.cynapsus.ca in the Investor Relations section.
About Cynapsus
Cynapsus is a specialty CNS pharmaceutical company developing and preparing to commercialize a fast-acting, easy-to-use, sublingual thin film for the on-demand management of debilitating OFF episodes associated with PD. The Company has successfully completed a Phase 2 clinical trial for its product candidate, APL-130277, a sublingual formulation of apomorphine hydrochloride, or apomorphine. Apomorphine is the only drug approved for acute, intermittent treatment of OFF episodes for advanced PD patients, but is currently only approved as a subcutaneous injection in the United States. APL-130277 is a “turning ON” medication designed to rapidly, safely and reliably convert a PD patient from the OFF to the ON state while avoiding many of the issues associated with subcutaneous delivery of apomorphine. It is designed to convert all types of OFF episodes, including morning OFF episodes, often considered the most difficult to treat. Cynapsus has initiated its Phase 3 clinical program for APL-130277, relying on the abbreviated Section 505(b)(2) regulatory pathway in the United States, and the Company intends to submit an NDA in the first half of 2017. For additional company information, please visit our website www.cynapsus.ca. For more information about the Phase 3 studies, including enrollment criteria, please visit the website found here http://cth300and301trials.cynapsus.ca/
Forward-Looking Statements
This announcement contains "forward-looking statements" within the meaning of applicable securities laws, including, without limitation, the Company’s expectation for filing an NDA in the first half of 2017; expectations regarding the Company’s clinical and regulatory activities, including without limitation, the anticipated timing, completion and results of Phase 3 and other clinical studies; and beliefs related to potential benefits, effectiveness and demand for, the Company’s product candidate. These forward-looking statements include information about possible or assumed future results of the Company’s business, financial condition, results of operations, liquidity, plans and objectives. In some cases, you can identify forward-looking statements by terminology such as “believe,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “expect,” “predict,” “potential,” or the negative of these terms or other similar expressions. These forward-looking statements are based on the Company’s current expectations and beliefs and inherently involve significant risks and uncertainties. Actual results and the timing of events could differ from those anticipated in such forward-looking statements as a result of risks and uncertainties, and include, but are not limited to, those factors identified under the caption “Risk Factors” in the Company’s Form 10-Q for the quarter ended March 31, 2022 filed with the United States Securities and Exchange Commission (the “SEC”) on May 11, 2016, and its other filings and reports in the United States with the SEC available on the SEC’s web site at www.sec.gov, and in Canada with the various Canadian securities regulators, which are available online at www.sedar.com. Furthermore, unless otherwise stated, the forward-looking statements contained in this press release are made as of the date of this press release, and the Company has no intention and undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events, changes or otherwise, except as required by law.
Neither the NASDAQ nor the TSX has approved or disapproved of the contents of this press release.
Contact Information Company Contact: Kristen Galfetti Vice President, Investor Relations (416) 703-2449 x246 [email protected] Media Contact: Russo Partners LLC Matt Middleman (212) 845-4272 [email protected]Source: Cynapsus Therapeutics Inc.
Released July 18, 2022